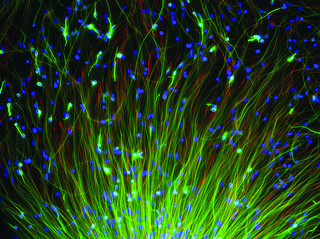
Transplant stat. Emory University School of Medicine neurosurgeon Nicholas Boulis MD injected between 0.5 and 1 million neural stem cells (above) in the lumbar section of ALS patients' spinal cords. Image: Corey Seehus, Brain Cells Inc for the 2010 GE Healthcare Image Competition.
Researchers estimate that more than 70% of motor neurons could be lost in people with ALS just one year after being diagnosed with the disease. To stem the tide of neurodegeneration, scientists are working hard to develop neuroprotective therapies to help keep their muscles moving.
One such potential treatment strategy, being developed by Maryland’s Neuralstem Inc., hopes to deploy neuronal bodyguards into the spinal cord that crank out protective substances which may shield the motor nerves from further destruction. The experimental stem cell transplantation procedure, performed during surgery, involves the direct injection of healthy neural stem cells into the spinal cord. The transplanted stem cells according to preclinical studies may give rise to populations of so-called interneurons that might plug directly into ALS-ravaged motor neurons, providing them life support.
In 2010, US physicians launched a phase I clinical trial to evaluate the safety of Neuralstem’s stem cell transplantation procedure. Patients received stem cell injections in the lumbar (leg-moving) region of the spinal cord. 12 people with ALS participated.
Now, the team reports the first results from the phase I clinical trial.
The stem cell transplantation procedure appears to be safe and does not appear to aggravate the disease. But, the immunosuppressants prescribed to prevent rejection of these transplanted stem cells were not well-tolerated by trial participants.
The results are published in the journal Stem Cells.
Emory University School of Medicine neurosurgeon Nicholas Boulis MD injected up to 1 million neural stem cells into the spinal cords of 12 ALS patients post-laminectomy. Patients received anti-rejection medicines routinely used in whole organ transplants before and after surgery.
The team found that the procedure appeared to be safe and 6 to 18 months later did not rapidly worsen patients’ condition according to ALS-FRS, forced vital capacity and other functional measures.
But researchers discovered that ALS patients could not tolerate the immunosuppressants (mycophenolate mofetil and tacrolimus) prescribed post-surgery. Most trial participants had to drop or reduce the dosage of at least one of the two anti-rejection medicines due to chronic bloating, diarrhea and vomiting. And, 2 out of 12 patients had to be taken off both drugs altogether due to the inability to control these symptoms through other medications.
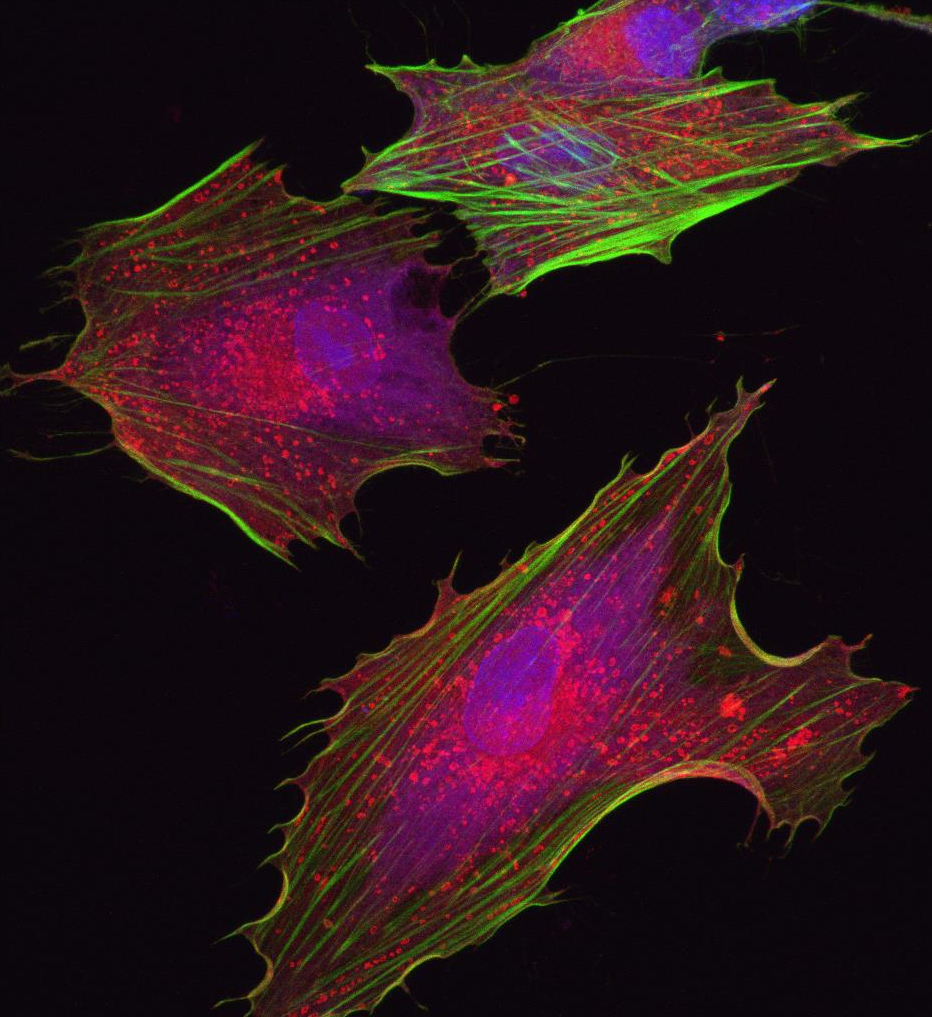
Under adult supervision. Scientists are creating potentially neuroprotective astrocyte-like cells from adult (mesenchymal) stem cells obtained from the patient's own bone marrow in hopes to slow down the disease. Image: Stem Cell Institute, Panama.
The jury is still out however whether a personalized stem cell therapy such as Brainstorm Cell Therapeutics’ NurOwn is a better way to go – particularly for people with inherited forms of ALS. The treatment strategy, which uses the patients’ own bone marrow to generate neurotrophin-producing cells, eliminates the need for immunosuppressants. But these transplanted cells according to some experts contain potentially ALS-triggering mutations which might also further contribute to the disease.
Neuralstem’s phase I clinical trial remains ongoing. Six people with ALS are expected to be implanted with stem cells in the cervical (diaphragm-moving) region of the spinal cord sometime this spring or summer. The trial is expected to be completed in October 2012.
Brainstorm Cell Therapeutics’ NurOwn phase I/II clinical trial is currently ongoing in Israel. Researchers are expected to launch a phase I/II ALS clinical trial in the US sometime in 2012.
References
Glass, J.D., Boulis, N.M., Johe, K., Rutkove, S.B., Federici, T., Polak, R., Kelly, C. and Feldman, E.L.(2012) Lumbar intraspinal injection of neural stem cells in patients with ALS: results of a phase I trial in 12 patients. Stem Cells, doi:10.1022/ stem.1079. Abstract | Full Text (Subscription Required)
Xu, L., Ryugo, D.K., Pongastaporn, T., Johe, K. and Koliatsos, V.E. (2009) Human neural stem grafts in the spinal cord of SOD1 transgenic rats: differentiation and structural integration into the segmental motor circuitry. Journal of Comparative Neurology 514(4), 297-309. Abstract | Full Text
Xu, L., Yan, J., Chen, D. Welsh, A.M., Hazel, T., Johe, K., Hatfield, G. and Koliatsos, V.E. (2006) Human neural stem cell grafts amerliorate motor neuron disease in SOD-1 transgenic rats. Transplantation 82(7), 865-875. Abstract | Full Text (Subscription Required)
Further reading
Boulis, N.M., Federici, T., Glass, J.D., Lunn, J.S., Sakowski, S.A. and Feldman, E.L. (2012) Translational stem cell therapy for amyotrophic lateral sclerosis. Nature Reviews Neurology 8(3), 172-176. Abstract | Full Text (Subscription Required)
Maragakis N.J. (2010). Stem cells for the neurologist. Amyotrophic Lateral Sclerosis, 11(5), 417-423. Abstract| Full Text (Subscription Required)
Patient Resources
Human Spinal Cord Derived Neural Stem Cell Transplantation for the Treatment of Amyotrophic Lateral Sclerosis (ALS) ALSTDI | Website | Contact
Autologous Cultured Mesenchymal Bone Marrow Stromal Cells Secreting Neurotrophic Factors (MSC-NTF), in ALS Patients. ALSTDI | Website | Contact
Note: We will update this information when details in regards to Brainstorm's US trial become available.